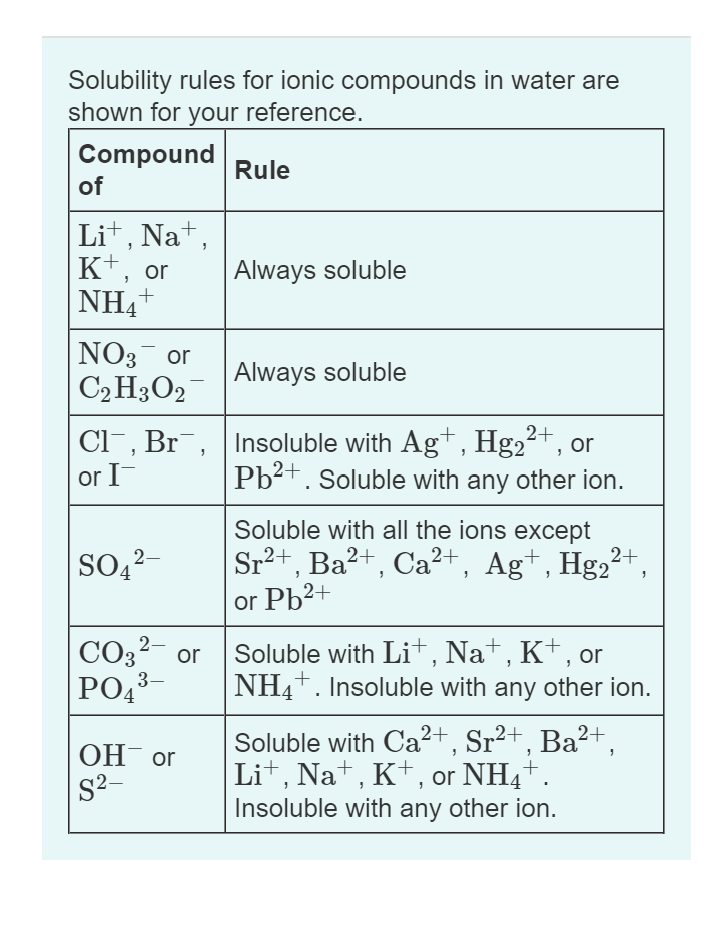
We will see later that there is an equilibrium between dissolution and precipitation, and the position of this equilibrium dictates whether we call a salt “soluble” or “insoluble.” The line is not perfectly sharp, which is why terms like “sparingly soluble” exist. The solubility of barium sulfate, a salt traditionally called insoluble, is 0.0002448 grams per 100 grams of water. calculate the solubility product, Ksp, given the solubilities and vice versa. For example, the solubility of glucose in water is 133 grams per liter. use the solubility rules to predict solubility behaviour of binary compounds. This concentration is the technical definition of solubility. We can write the saturation point as the maximum concentration of a solute in water. From a microscopic perspective, every solute is “soluble” in pure water! In fact, one way to think of insoluble species is as soluble species with very small saturation points. This point of saturation is different for different solutes. Only those metal ions that form very insoluble sulfides, such as As 3+, Bi 3+, Cd 2+, Cu 2+, Hg 2+, Sb 3+, and Sn 2+, precipitate as their sulfide salts under these acidic conditions. Group 2: Acid-Insoluble Sulfides Next, the acidic solution is saturated with H 2 S gas. Solubility is not a cut-and-dry concept: at some point, when a solution has become saturated with the cation and anion contained in an ionic salt (or with a covalent solute), no more solute can enter the solution. The precipitate can be collected by filtration or centrifugation. Acetates and nitrates are cleanly soluble, while salts of other polyatomic ions are spotty.ĭo not memorize the solubility rules they’ll be provided where you need them. What are the trends here? Generally, salts of the extremes on the periodic table (group 1 cations and group 17 anions) are soluble. Carbonates (CO 3 2–), hydroxides (OH –), oxides (O 2–), and phosphates (PO 4 3–) are insoluble, except for salts of group 1 cations.Sulfates (SO 4 2–) are soluble with the exception of salts of barium, strontium, lead, calcium, silver, and mercury(I).All chlorides, bromides, and iodides are soluble with the exception of salts of silver, lead, and mercury(I).the net ionic equation for the formation of Cus would show only these two ions. The formula of copper (II) sulfide is CuS. For each reaction name the appropriate spectator ions. All common acetates (CH 3CO 2 –) and nitrates (NO 3 –) are soluble. Write the net ionic equation for the formation of a precipitate of calcium phosphate.Salts of group 1 cations (Li +, Na +, and K +) are soluble.Here are the solubility rules in all their glory, listed roughly in order of importance. We’ll leave aside for now what exactly we mean by “soluble”… By determining how the rules apply to a given salt, we can predict whether it will be soluble in water or not. The solubility rules speak to the solubility of salts of cations within a particular group (e.g., group 1 cations) or specific anions. Over the years, solubility rules have been developed to help us predict whether a given salt will be soluble in water. When the ionic bonds are especially strong, however, solvation may not be thermodynamically favored! Not all ionic solutes are soluble in water. This article was written for you by Jeremie, one of the tutors with Test Prep Academy.In the last section, we saw that ionic solutes dissolve in water when ion-dipole forces overwhelm the ionic bonds in the solid solute. Looking to get ready for the ACT? We can help with ACT Prep 
No other rule above 5 applies to this compound and so rule 5 states that hydroxides are insoluble. An additional upgrade, Violent Precipitate Set Style Unlock, can be found along the way. Even though rule 5 states that carbonates are insoluble, rule 1 is higher and thus this compound is soluble in water. Lantern of the Violent Precipitate Helm of the Violent Precipitate Bracers of the Violent Precipitate Crystals of the Violent Precipitate This set is awarded to players who reach the end of the Cavern Crawl utility path. Rule 2 states that all nitrates are soluble. Rule 5: Carbonates, oxides, hydroxides, and phosphates are insoluble. All common salts of the Group 1 elements and the ammonium ion are soluble. Rule 4: All sulphates are soluble except those with barium, strontium, lead, calcium, silver and mercury. Rule 3: All compounds with halogens and a metal except for fluorine are soluble except those with silver, mercury and lead. Rule 2: All nitrates and acetates are soluble. Rule 1: All salts that include a group 1 element or ammonium are soluble. The rules are listed in order of importance and rule 1 will overwrite rule 5. There are set of rules that are a guide to determine if an inorganic compound is soluble in water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
